
Discover key steps for equipment qualification of sterile fill line equipment, ensuring FDA compliance with best practices from installation to performance qualification.

Prevent FDA Form 483 issues with expert strategies in compliance, data integrity, and quality systems from cGMP Consulting.

Ensure data integrity during Commissioning, Qualification, and Validation (CQV) with proven practices from cGMP Consulting. Learn how we address FDA concerns, implement automated systems, and follow ALCOA+ principles to maintain data accuracy and compliance in equipment qualification.

Uncover the financial and operational risks of an FDA Form 483, from costly remediation to reputation damage. Learn how cGMP Consulting can help mitigate these impacts and ensure compliance.

Step-by-step guide to equipment qualification for FDA compliance, covering Installation, Operational, and Performance Qualification (IQ, OQ, PQ) and maintaining product quality in new facilities.

Learn the key steps to take after receiving an FDA Form 483 to avoid a Warning Letter, including swift response, CAPA plan development, and maintaining FDA communication.

Prepare your organization for success with cGMP Consulting's Audit Readiness Training. Our 5-step process equips your team to excel in regulatory inspections.

Learn about Commissioning, Qualification, and Validation (CQV) in cGMP environments to ensure compliance and product quality in the pharmaceutical industry.

Prepare for MoCRA GMP compliance with cGMP Consulting. Get expert help with assessments, SOPs, and training for early compliance and reduced risks.

Discover how to seamlessly implement electronic GMP systems with expert consulting from cGMP Consulting and amni.ai. Enhance compliance and operational efficiency in FDA-regulated industries.

Explore the essential elements of cosmetic GMPs and learn how cGMP Consulting can help ensure the safety, efficacy, and quality of your cosmetic products while navigating regulatory compliance.
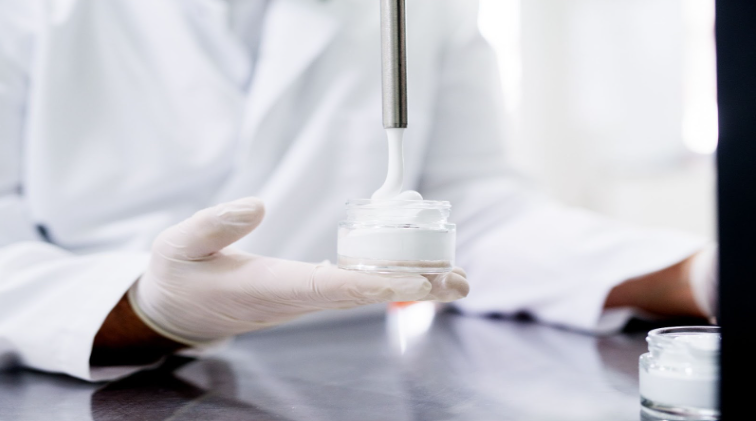
Simplify MoCRA compliance with cGMP Consulting. Our guide covers key elements like GMP implementation, safety substantiation, labeling, adverse event reporting, and more.

Master the GMP certification process with cGMP Consulting’s ultimate guide. Learn about our comprehensive evaluation, certification recommendations, and ongoing compliance support.

Discover the 8 key challenges in obtaining and maintaining GMP certification and how cGMP Consulting can help your organization achieve compliance and improve product quality.

Elevate your quality standards with cGMP Consulting's GMP Certification Audits. ISO/IEC 17020:2012 accredited, we offer tailored audits for compliance and quality assurance.

Discover the benefits and challenges of assisted line clearance in manufacturing. Learn how automation can enhance efficiency, quality, and compliance.

Explore the benefits of Vaporized Hydrogen Peroxide (H2O2) Pass-Through Chambers for cleanroom decontamination. Learn how VHP effectively bio-decontaminates materials while meeting ISO standards.

Master GMP compliance with cGMP Consulting's comprehensive auditing services. Explore 8 tailored solutions to enhance product quality, ensure regulatory adherence, and build market trust.

Explore the importance of effective technical writing in deviation management for FDA-regulated industries. Learn how cGMP Consulting can enhance compliance and ensure patient safety.

Discover effective strategies for managing deviations in regulated industries. Learn how cGMP Consulting can help improve compliance and operational efficiency.

Learn how to conduct effective cross contamination risk assessments to manage contamination risks in production processes.

Explore effective risk management strategies in regulated industries. Learn how cGMP Consulting can enhance your risk assessment processes for safety and compliance.

Explore glove testing methods in drug manufacturing to ensure contamination prevention and compliance with industry standards.

Explore the Factory of the Future with Shivani Upadhyay’s guide. Learn about digital technologies, agile processes, and modular tech to transform your manufacturing.

Learn how to create an effective Periodic Validation Review (PVR) Schedule for qualified equipment and systems

Discover what a Periodic Validation Review (PVR) is, how it's conducted, and how often it should be performed to ensure ongoing compliance and system control.

Learn how pharmaceutical aggregation establishes traceability in the supply chain by linking serialized packaging.

Temperature mapping is a Good Manufacturing Practice (GMP) utilized by a variety of industries to capture how temperature is distributed within a space.

Cleanrooms are highly controlled spaces where pharmaceuticals are produced. Cleaning is the key element of contamination control.

Automated cleaning in pharmaceuticals optimizes labor efficiency, ensures consistent results, and enhances safety through PLC monitoring.



